Biotech & Pharma
France: A European Leader in Biotherapeutics and Biomanufacturing
France has emerged as one of the most dynamic and integrated global hubs for biotechnology and pharmaceuticals. The country combines world‑class science, advanced industrial capabilities, major public investments, and a highly structured ecosystem centered on innovation and health sovereignty.
With a mature R&D landscape, a strong clinical network, expanding biomanufacturing capacity, and a robust industrial base, France offers an unparalleled environment to innovate, scale, and commercialize next‑generation therapies.
A Fully Integrated Ecosystem: From Discovery to Patients
France’s competitive advantage lies in its unique ability to connect fundamental research, clinical development, industrial scale‑up, and market access — all within a single, coordinated ecosystem.
Scientific Excellence and Early‑Stage Innovation
France hosts:
- internationally recognized research institutions (Inserm, CNRS, CEA),
- high-impact academic labs,
- strong interdisciplinary collaboration,
- a dense network of startups and deeptech ventures.
This scientific foundation fuels a continuous flow of innovative therapeutic candidates and disruptive technologies.
Clinical Research Excellence
Clinical Research Excellence
France is one of Europe’s leading centers for clinical research, supported by:
- a broad network of academic hospitals and specialized centers,
- high patient enrollment capacity,
- proven excellence in oncology, immunology, rare diseases, infectious diseases, neurology, and metabolic disorders.
Industrial Strength and Regulatory Expertise
Major pharmaceutical groups — French and international — rely on France for:
- development and regulatory excellence,
- experienced talent pools,
- robust supply chains,
- advanced production sites,
- competitive operating conditions.
This continuum positions France as one of the most complete, high‑performance life sciences ecosystems in Europe.
Biomanufacturing: A Strategic Pillar and Competitive Edge
Biomanufacturing stands at the heart of France’s long‑term strategy. The ambition is clear: to make France a global reference for the industrial production of biologics and advanced therapies.
Massive Investment and Rapid Capacity Expansion
Under Innovation Santé 2030 and the France 2030 national plan, France is accelerating the development of:
- GMP production infrastructures,
- cutting‑edge bioproduction platforms,
- fill‑finish capacities,
- scale‑up and technology transfer programs,
- ATMP‑ready facilities.
These investments reinforce France’s industrial resilience and its ability to host major international bioproduction projects.
An End‑to‑End Bioproduction Value Chain
France offers a complete industrial pathway, including:
- Upstream processing: cell engineering, seed train, bioreactors
- Downstream processing: capture, purification, formulation
- Quality & release: analytics, QC, GMP compliance
- Fill‑finish and packaging
This integrated model ensures quality, reliability, scalability, and time‑to‑market performance, all critical for global biopharma companies.
A Collaborative Ecosystem Built for Partnership and Acceleration
Collaboration is a defining feature of the French health innovation model. The ecosystem brings together:
- national authorities and health agencies,
- hospitals and research centers,
- scientific and clinical experts,
- pharmaceutical companies and biotechs,
- investors and industrial partners,
- leading clusters and bioparks.
This culture of partnership enables faster innovation cycles, smoother regulatory pathways, and high‑value industrial and clinical collaborations.
National Clusters and Centers of Excellence
France is home to:
- major pharmaceutical clusters,
- specialized bioclusters,
- world‑class research centers,
- industrial zones hosting tens of thousands of skilled jobs,
- advanced clinical infrastructures.
Recent national announcements include the creation of 12 new University Hospital Institutes (IHUs) and 4 new bioclusters, further strengthening excellence in translational research and therapeutic innovation.
A Growing, Resilient, and Highly Attractive Industry
Despite global market challenges, the French healthtech and biopharma sectors continue to demonstrate:
- strong resilience,
- consistent investment inflows,
- growth in employment (+18% in two years),
- intensified partnerships with global industry leaders.
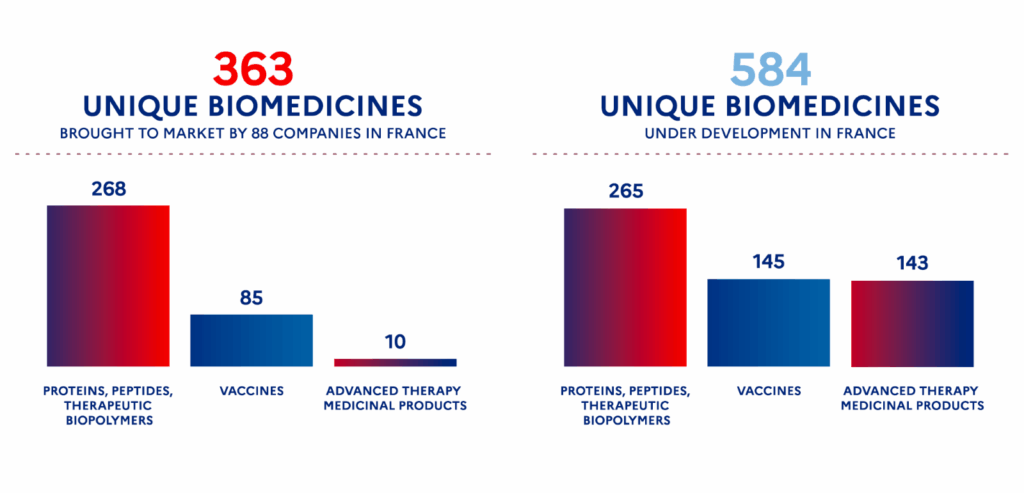
France now ranks among Europe’s top performers, with:
- the 2nd largest biopharmaceutical pipeline in Europe,
- a leading position in rare diseases and advanced therapies,
- one of the highest volumes of venture capital investment in healthtech,
- a strong export footprint — pharmaceuticals are one of France’s rare industries with a positive trade balance.
Its companies maintain an active presence in both Europe and the United States, demonstrating the ecosystem’s maturity and global reach.
France 2030: Accelerating Innovation and Industrial Sovereignty
France 2030 is one of the world’s most ambitious national strategies for health innovation and biomanufacturing.
Strategic Priorities
- accelerate development of breakthrough therapies,
- strengthen translational and clinical research,
- support industrial scale‑up and local manufacturing,
- secure strategic supply chains,
- enhance access to innovation for patients,
- reinforce national and European sovereignty.
More than 19 of the 21 measures under Innovation Santé 2030 have already been implemented, resulting in:
- €338M invested in R&D and therapeutic innovation projects,
- €50M dedicated to industrialization programs,
- €80M for biomanufacturing research,
- over 80 funded projects involving more than 250 partners nationwide.
This long‑term vision is transforming France into a major global player in biomedicine and biomanufacturing.
A Prime Destination for Global Investors
France is today:
- the leading European destination for health manufacturing projects,
- a highly competitive location for industrial investments,
- a strategic entry point to the European market,
- a country with a stable business environment and a skilled workforce.
Global companies choose France for:
- the quality of its scientific and clinical ecosystem,
- strong public support mechanisms,
- business‑friendly policies for innovation,
- access to key markets,
- cost‑competitive industrial operations,
- reduced time‑to‑market for innovative therapies.
From R&D to large‑scale production, France offers excellent conditions for long‑term growth.
Business France: Your Partner for Growth and Investment
Business France supports international companies and investors across the entire project lifecycle:
Invest & industrialize
- site selection and feasibility assessments,
- industrial implantation, expansion, or scale‑up,
- ATMP and bioproduction‑ready sites.
Innovate & collaborate
- connection to research centers, hospitals, clusters, and scientific partners,
- support for R&D partnerships,
- navigation of incentives and public funding programs.
Export & expand
- international development strategies,
- access to global markets and ecosystems.
Business France brings:
- deep sector expertise,
- a nationwide and international network,
- personalized, confidential assistance,
- support designed to accelerate project execution.
Business France’s mission: help you succeed in France and scale globally.
Conclusion
France stands among the world’s leading destinations for pharmaceutical innovation, biotechnology, and biomanufacturing. Its integrated ecosystem — combining scientific excellence, clinical strength, industrial capability, and strong public support — creates ideal conditions for developing and producing the therapies of tomorrow.
For companies seeking to innovate, manufacture, partner, or invest, France offers a unique, competitive, and forward‑looking environment.

